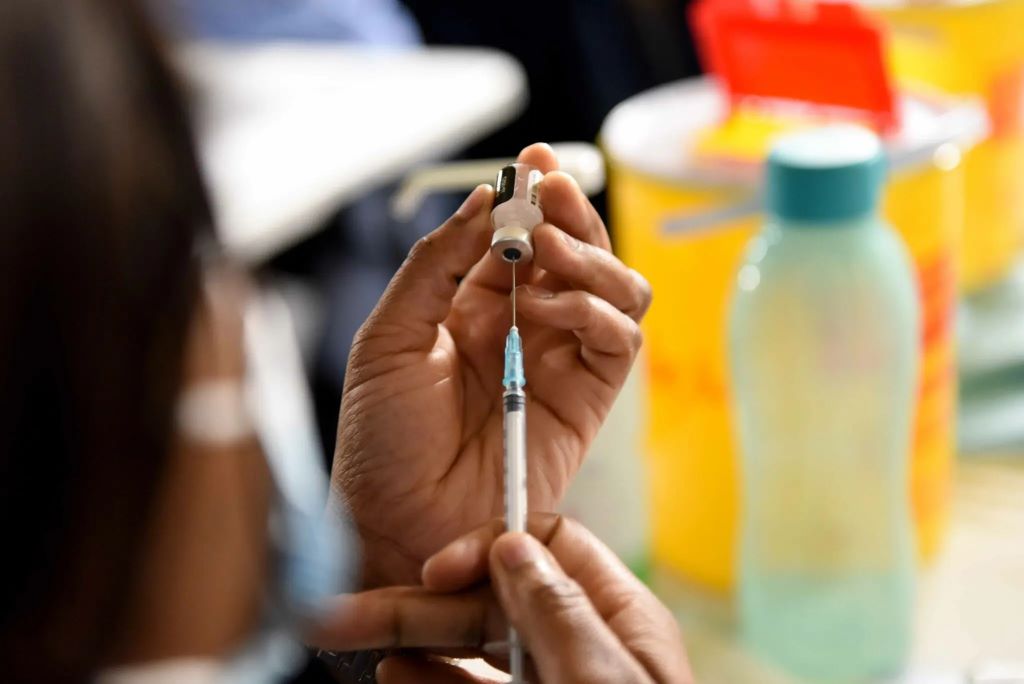
A new HIV prevention injection is now available to a select number of people in South Africa. That a single shot provides two months of protection is one of the injection’s major selling points. In this story, Elri Voigt unpacks how much of the jab is available, who is choosing to get it and what other anti-HIV drugs are being rolled out.
By Elri Voigt for Spotlight
Earlier this month, a young person in Cape Town became one of the first people in the country to receive a new HIV prevention injection outside of a clinical trial. The injection contains a long-acting formulation of the antiretroviral drug cabotegravir (CAB-LA for short). It provides two months of protection against HIV infection per shot.
“We were excited and nervous at the same time because (we) didn’t know how this person is going to react to an injection,” said Pakama Mapukata, a nurse and study coordinator. She added that the first person who received the CAB-LA injection responded well and told her that the injection was less painful than an sexually transmitted infection (STI) injection they had to receive in the past.
While the injection is not readily available for most members of the public just yet, a select number of people in the country will be able to access it via several implementation studies, also called pilot projects. One of these pilots is a study called FAST PrEP, conducted by the Desmond Tutu Health Foundation (DTHF) in Cape Town. Technically, access to the injection is limited to a FAST PrEP sub study called Prepare to Choose.
Taking antiretrovirals to prevent HIV infection is referred to as pre-exposure prophylaxis (PrEP). PrEP is available in the form of pills, vaginal rings, and injections.
According to Elzette Rousseau, a social behavioural scientist and the lead co-investigator in the implementation team for FAST PrEP, on the first day it was offered, five people opted to get the CAB-LA shot. “The first two, at least, that came through was a young MSM [men who have sex with men] and one was a young woman, which is definitely exciting because that is the population that we would want to come to our services which will benefit most from it,” she said. As of 21 February, 19 injections in total had been administered.
‘Real-world experience’
Professor Linda-Gail Bekker, Chief Executive Officer of the DTHF and Principal Investigator of the study, explained that once CAB-LA demonstrated efficacy in phase three clinical trials, it was decided to first do some implementation science studies in the country, alongside the other new PrEP option which is the dapivirine vaginal ring (DPV-VR), before rolling it out in the public sector.
Both the CAB-LA injection and the dapivirine ring have been approved by the South African Health Products Regulatory Authority (SAHPRA). Prevention pills, also called oral PrEP, were approved several years earlier and are already widely available in the public sector and at pharmacies.
She explained the idea is that these implementation studies can help transition the product from the clinical trial setting to a real-world rollout in the public sector. Essentially the pilots would serve as a way of introducing the injectable and the ring on a smaller scale and lessons learnt from the pilots could be used to inform the future, larger rollout of these products. It also helps pick up any potential issues or safety concerns that may not have been seen in the clinical trials.
She added that pilot projects also help inform what the demand for a product like CAB-LA and the DPV-VR will be, which can help with advocacy efforts and give the manufacturers and companies who create generic products an idea of whether it’s worth investing in these products.
“There really are limited pilots going on in the country to date,” Bekker said. The pilots that are offering CAB-LA in addition to the DTHF are being conducted by Ezintsha and Africa Health Research Institute (AHRI), as well as the Wits Reproductive Health and HIV Institute (Wits RHI). Spotlight reported on this in-depth last year.
CAB-LA delays
Bekker told Spotlight the volumes of CAB-LA available in the country remain constrained for now.
While SAHPRA approved the injection in late 2022, limited supply and the product’s high price has limited uptake around the world. A recent HIV investment case for South Africa found the injection not to be cost-effective at the current price compared to PrEP in the form of pills. For now, the only supplier of CAB-LA is the pharmaceutical company ViiV Healthcare. Generic products are anticipated to enter the market in three to four years.
Despite SAHPRA approval for the product, the pilot projects have experienced delays in getting CAB-LA to their participants. As Spotlight reported last year, the National Department of Health stated that there were challenges getting the CAB-LA injections donated for the implementation studies into the country as the packaging did not meet South African regulatory requirements.
Bekker said that an alternative is to import CAB-LA through a phase 3b study (in this case the Prepare to Choose study), approved by SAHPRA’s Clinical Trial committee. Writing up protocols and having the study approved by an ethics committee and SAHPRA took some time, and once it was approved, CAB-LA still needed to be imported and ViiV Healthcare had to ramp up manufacturing to meet demand.
Bekker told Spotlight that to date, CAB-LA has not yet been purchased by the National Department of Health for distribution to the public, and the only other way to get CAB-LA into the country will be through a donation by the United States President’s Emergency Plan for AIDS Relief (PEPFAR).
“PEPFAR has been able to import the product into Zambia and Malawi…as the first two PEPFAR countries to get it as a PEPFAR donated public rollout and we hope South Africa is in that queue further down the line,” she said.
The Prepare to Choose Study
At the moment, Prepare to Choose can only offer CAB-LA to a few hundred people. Bekker said that ideally, they would have wanted to offer all their FAST PrEP clients a three-way choice of either the vaginal ring, oral PrEP pills or CAB-LA. But for now, CAB-LA is only being offered within Prepare to Choose, which is a single-nested sub study within FAST PrEP.
Mapukata, who was present during the first CAB-LA injection in the implementation study, said it will be interesting to see what participants choose now that they have an additional PrEP option. “People have been waiting for injection for the longest time, so we are seeing lots of excitement from the participant side,” she said.
Rousseau told Spotlight that Prepare to Choose currently has enough CAB-LA doses for 900 participants over an 18-month period.
She said they have thus far observed that “people are still choosing what [PrEP option] suits them” when offering existing or potential FAST PrEP participants the choice to access CAB-LA.
So far those who have chosen CAB-LA are primarily adolescent girls and young women with an average age of 22. Some have been on PrEP before, while others are starting PrEP for the first time. “In that cohort we know that the burden of HIV exists, so that’s encouraging at this point,” Rousseau said.
Trends observed in FAST PrEP
FAST PrEP is being implemented at 12 public sector health facilities in the Klipfontein and Mitchells Plain Health Sub-Districts in the Western Cape, as well as in four mobile clinics that operate in the area. Since the start of FAST PrEP, just under 11 000 participants have enrolled, according to Rousseau. This means that around 11 000 people have accessed either prevention pills or the DPV-VR through the study.
When FAST PrEP started, the assumption was that the study can enrol between 20 000 and 23 000 participants, but it is not necessarily targeting to enrol that exact number of participants. Rousseau added that the study currently has funding to continue offering PrEP until late next year but access to these options may potentially continue beyond that.
The study reaches participants in public sector healthcare facilities by having two peer navigators in each facility. These peer navigators are young people trained and employed by the study coordinators. They can educate and counsel young people about FAST PrEP. The study coordinators also offer training, particularly sensitisation training, to nurses and other staff members.
The four mobile clinics travel around the Klipfontein and Mitchells Plain Health Sub-Districts, particularly where there is a high incidence of HIV, as well as spaces where young people are present. These include 16 secondary schools in the area where the mobile clinics have permission to enter the school grounds.
Demand for the DPV-VR
Rousseau told Spotlight that so far, just under 200 women in the study have chosen to use the DPV-VR. However, it’s important to note that within the whole study population, not everyone is eligible to use the ring. It is currently being offered to women who are over 18, not pregnant and not breastfeeding.
She added that for participants who are eligible for both the ring and oral PrEP, the pill is still more popular – with a rough estimate of around 15% of eligible participants opting for the ring. Most participants, at this stage, who choose to use the ring are those who have tried oral PrEP first and struggle to take pills daily or found it doesn’t suit their lifestyle. Very few participants to date have started on the ring and then switched to the daily pill.

She said the demographics of who prefers the ring over oral PrEP haven’t been explored in-depth, but it’s something that the study will be looking at and analysing data on in future.
Bekker added to this saying: “We always expected it to be a bit of a niche product because you know definitely for many the idea of swallowing a pill is perhaps an easier concept than using a vaginal ring. So, it has started slowly, we’ve now administered hundreds as opposed to thousands of rings.”
She noted that interest in the ring has built overtime and is starting to pick up more. “Our first, preliminary data suggests that the women who choose rings are coming back [for it] …they’ve decided they want to go that road and they’ve committed,” Bekker said.
Counselling for Choice
While the ring was found to be effective in two phase 3 trials, its efficacy in those trials was far from 100% and the evidence for the ring’s efficacy is generally less impressive than that for pills and the injection. Interpreting findings from PrEP trials is also somewhat muddied by whether or not pills are taken as prescribed, and the ring is used and replaced as prescribed – that a single shot provides two months of protection is one of the injection’s major selling points.
Compared to placebo, there was a 30% reduction in HIV infection for ring users in phase three trials, while there was a 50 to 60% reduction in infection when the ring went to open-label, Bekker noted.
She said that it has previously been observed that clinical trial efficacy results can differ from real-world results, particularly when it comes to HIV prevention. For instance, she said, oral PrEP in clinical trials initially showed no evidence of efficacy in the prevention of HIV in women. Yet, real-world evidence showed it works in all populations if taken as prescribed.
What both these cases have shown, according to Bekker, is that it’s not necessarily that the product isn’t working, it’s that the product isn’t always being used as intended. When it comes to the ring, she said, the drug within the ring is efficacious and will kill the virus, but the ring must be present at the time that the individual is exposed to HIV. “Once you take the ring out, the [prevention] effect is lost,” she said.
When asked how women are counselled about the ring in the FAST PrEP study, Bekker said it is done very carefully and with guidance of their peers – this is where the peer navigators play a big role.
FAST PrEP was designed using a lot of engagement from young people, Bekker said. For a year before the pilot started, a group of 100 young people from diverse populations were enrolled from the community to give feedback on how to design the pilot so it can best reach young people. This group also essentially helped troubleshoot the information coming from the pilot to ensure that the PrEP choices were communicated in an appropriate way.
“They are very instrumental at the moment in making sure that that message [on DPV-VR] is clearly communicated,” she said.
Bekker added that if an individual needs time to think about which PrEP option to use, they are advised to start with oral PrEP and that they can switch later if they want.
Mapukata explained how the counselling process plays out on the ground. Participants in FAST PrEP, once they have spoken to a peer navigator, are taken into a counselling room and given a quiz where their scores are used to indicate what PrEP option might work for them. This is used as a starting point to counsel participants about the different PrEP options and which options they are eligible for and most comfortable using.
“It’s a lot of counselling that goes in before that choice [of PrEP] is made,” Mapukata said.
Young people who are members of the FAST PrEP youth reference group speak of the project in glowing terms. “And it’s so nice because you have a variety to choose from, you’re not obligated [to only] be on PrEP, on the oral, because there’s a variety of options,” one of them told Spotlight.
Republished from Spotlight under a Creative Commons licence.
Source: Spotlight

