Head Cooling May Temporarily Relieve Depression Symptoms
People who wore a cooling cap for 30 minutes experienced multiple changes that could affect their mental health, according to a pilot study by Penn State researchers
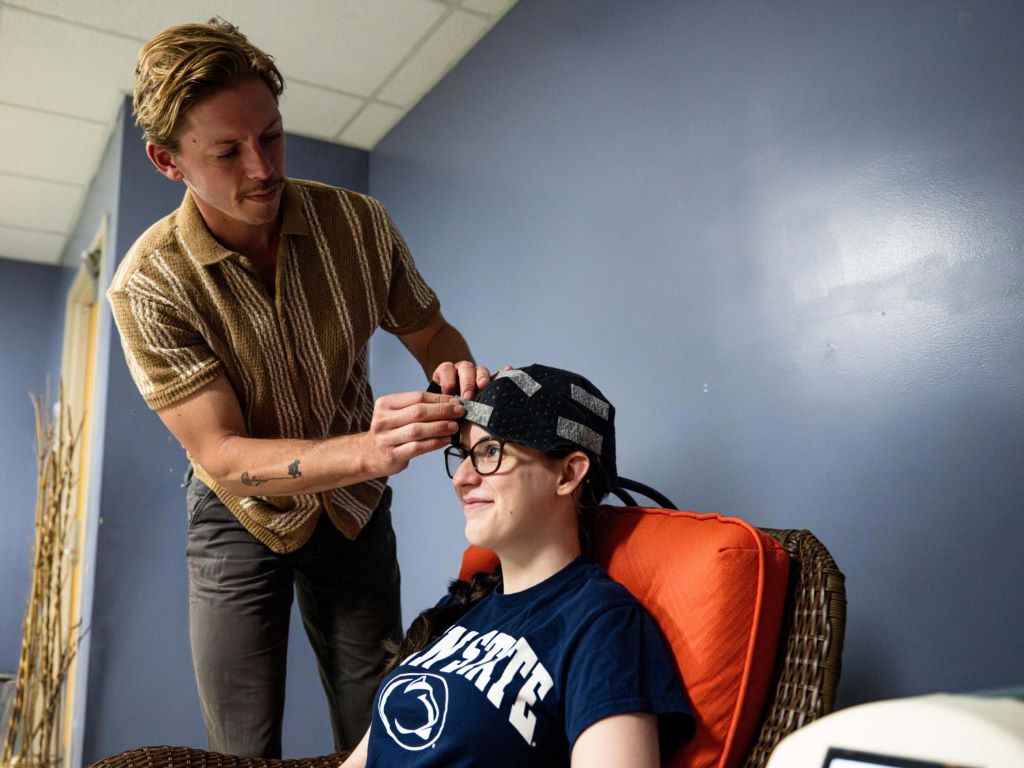
Co-authors Owen Griffith, standing, and Maddie McLaughlin demonstrate the head cooling cap used in the study. Credit: Jaydyn Isiminger / Penn State. Creative Commons
Wearing a cooling cap for 30 minutes may improve a person’s sense of well-being, according to a new study by Penn State researchers.
In a recent publication in Acta Psychologica, the researchers demonstrated that head cooling may reduce depressive symptoms and alter the types of brain waves people produce. While no medical recommendations can be derived from this small, exploratory study, the results indicate head cooling may provide mental health benefits for the general population.
The work was inspired by lead author and Penn State Professor of Kinesiology Semyon Slobounov’s prior research, which found that athletes with concussions heal faster and experience fewer symptoms when their head is regularly cooled.
“A person’s mood is tied to their cognition and general brain function,” said Owen Griffith, assistant teaching professor of kinesiology at Penn State and co-author of the study. “In this study, results suggested that people enjoy the sensation of head cooling. This, in turn, improved their mood, which altered their brain activity.”
The researchers recruited 24 college students between the ages of 18 and 26. At the beginning of the study, all participants completed questionnaires that measured their mental health and cognitive abilities and underwent an electroencephalogram (EEG) to measure brain activity.
Following the EEG, participants spent 30 minutes sitting in a dimly lit room listening to ocean sounds. Half of participants wore a fitted cooling cap, which uses liquid circulating close to the head to maintain a temperature of 33 degrees Fahrenheit. The other participants wore nothing on their heads. Immediately after the cooling or sitting session, participants repeated the questionnaires and EEG.
Participants repeated the same sitting or cooling session without testing every day for one week. The day after the last session, participants repeated the questionnaires and EEG again. This design allowed the researchers to observe both the short- and longer-term effects of head cooling.
“The brain produces different types of waves that are associated with different levels of excitement or brain activation,” said Laura Cooney, co-author of the study who graduated from Penn State’s Schreyer Honor College in 2025 and based her undergraduate thesis on the research. “Alpha waves are associated with calmness. More specifically, they are indicative of less brain activity overall, so this finding suggests that there was an immediate calming effect of head cooling.”
People in the head cooling group displayed an increase in alpha brain waves during the EEG immediately following the first cooling session. They experienced a 4% increase in alpha waves while participants whose heads were not cooled displayed a .5% decrease in alpha waves.
In contrast, there was no significant difference in the alpha wave levels of the sitting and cooling groups when measured on the day after the final cooling session, suggesting cooling does not have a longer-term impact on brain wave activity, the researchers said.
Over the course of the week, both groups of participants reported a decrease in depression symptoms, but individuals in the head cooling group reported a larger decrease than those in the sitting group.
“The reduction of depression symptoms among healthy people suggests that this might be a promising treatment,” Griffith said.
The researchers said they had hypothesised head cooling affected people through changes in neural electrical activity, but the EEGs did not show evidence of that. Now, the researchers suspect the effects are psychosomatic, meaning that mental and emotional factors, rather than physiological changes, are causing people’s reduced depression symptoms and increased alpha brain wave activity.
“Anecdotally, most people who come into the lab agree that head cooling is relaxing and enjoyable,” Griffith said. “This may not be surprising. A cold compress or a bag of ice have been home treatments for migraines for many years.”
Overall, the study suggests that widespread head cooling could be useful, the researchers said.
“Head cooling shows some potential as an acute calming therapy,” Cooney said. “Not as a replacement for any current therapy, but as another tool in the toolbox.”
Slobounov, senior author of the study, agreed.
“Our previous research demonstrated that head cooling is useful for athletes recovering from concussions,” Slobounov said. “This research suggests it may be more useful to a wide group of people. It is low risk, does not involve any drugs or chemicals, and people enjoy it.”
Other Penn State researchers who contributed to this work include Zach Napora, graduate student in kinesiology and first author of the publication, Maddie McLaughlin, graduate student in kinesiology, and Elle McNally, 2025 graduate in biobehavioral health and current physician assistant graduate student.
Source: Penn State University